PEI MAX® - Transfection Grade Linear Polyethylenimine Hydrochloride (MW 40,000)
Product Number:
CAS #
-
24765
-
49553-93-7
Product #
CAS #
-
24765
-
49553-93-7
Product Description
Polyethylenimine “Max” (PEI MAX) is a powerful, trusted, and cost-effective reagent widely considered as a current gold standard for both in vitro and in vivo transfection. PEI MAX has a high density of protonatable amino groups, with amino nitrogen as every third atom. This imparts a high buffering ability at nearly any pH. Hence, once inside the endosome, PEI MAX disrupts the vacuole and releases the genetic material into the cytoplasm. Stable complexation with DNA, efficient entry into the cell, and ability to escape the endosome makes PEI MAX a highly efficient transfection reagent which is compatible for a wide range of cell lines/types including the most commonly used HEK293 and CHO cells grown in adherent and suspension cultures. PEI MAX is capable of yielding high efficiency cell lines without compromising cell viability compared to other PEI and liposomal transfection reagents available in the market.
Key Advantages
- Superior Performance: High transfection efficiency with low cytotoxicity compared to other reagents on the market, suitable for use in larger concentrations and in sensitive cells.
- Low cytotoxicity: Even larger concentrations of PEI MAX disrupts cells minimally
- High Quality: Sourced and manufactured in the U.S. under ISO 13485 Quality System
- Flexible Workflow: Easy to optimize and introduce into application protocols. Scalable for well plates, flasks, and larger capacity bioreactors.
- Available in cGMP grade and in both powder and liquid formats.
- Cost-Effective: Economical compared to similar transfection products in the market.
- History: Polysciences is a trusted partner for pharma and medical device key players with more than 60 years in specialty chemical manufacturing. PEI MAX has been used by thousands of customers in their research and development for more than a decade.
POLYSCIENCES’ PEI MAX PRODUCT YIELDS HIGH TITERS OF AVV AFTER 72 HOURS POST TRANSFECTION
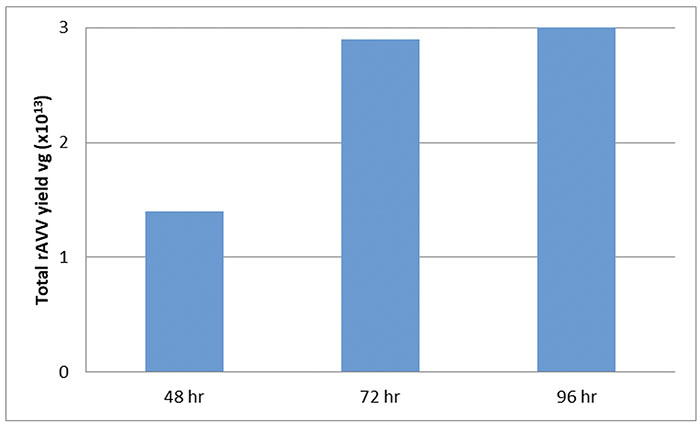
Flasks containing 30mL cultures of suspension HEK293 cells at 1×10^6 cells/mL were transfected to produce rAAV8 CMV-eGFP. rAAV8 was harvested from the media 48 hours post-transfection with increasing amounts of vector found in the media over time (Griger 2015).

